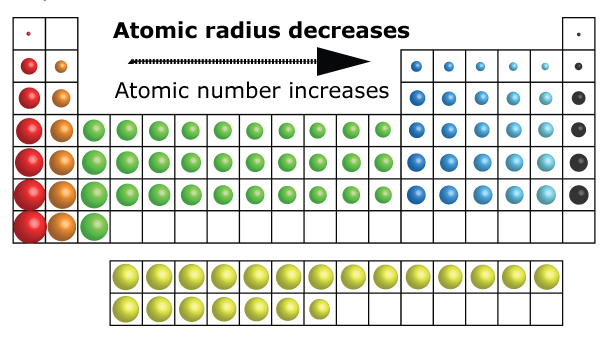
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals. Consequently, beryllium is significantly smaller than lithium. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. The covalent radii of the main group elements are given in the figure below. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a Cl 2 molecule. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. When an electron is added, a new proton is also added to the nucleus, which. 
Within a period of elements, each new electron is added to the same shell. The first atomic radius periodic trend is that atomic size decreases as you move left to right across a period. atomic size affects both electronegativity and polarization. Atomic Radius Trend 1: Atomic Radii Decrease From Left to Right Across a Period. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. Compounds that O forms with elements on the left - hand side of the. Because an atom does not have a discrete boundary, there are various non-equivalent definitions of atomic radius, such as Van der Waals radius and covalent radius. 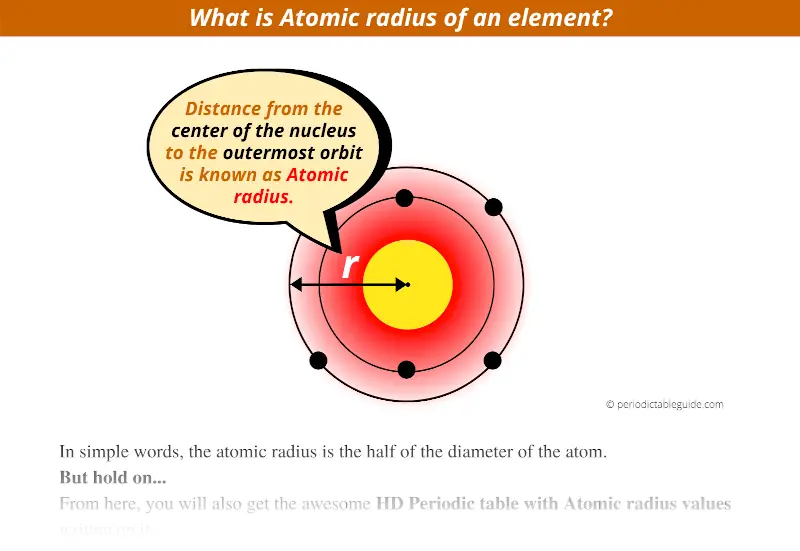
The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. The atomic radius of a chemical element is a measure of the size of its atom, usually, the distance from the center of the nucleus to the outermost isolated electron. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
